Transforming Immunotherapy through Granzyme B Insights[I]Forward-looking statements. Statements about potential benefits, future studies, timelines, or development plans are forward-looking and subject to risks and uncertainties; actual results may differ.
Granzyme B–guided insights from ongoing studies — where advanced radiodiagnostics meet targeted radiopharmaceuticals in development.[II]Early/ongoing human research. Human data referenced are from early, exploratory, or ongoing studies and may be subject to change as additional data become available.
Our Radiodiagnostic Data
We have radiodiagnostic clinical study readouts across melanoma, NSCLC, colitis, inflammatory lung disease, and combination-therapy cohorts using CSB-321 PET.[II]Early/ongoing human research. Human data referenced are from early, exploratory, or ongoing studies and may be subject to change as additional data become available. Findings include lesion-to-lesion heterogeneity and therapy response assessments in both cancer and autoimmune disease.[V]Lesion-level heterogeneity. Lesion-level readouts can vary within the same patient; single-lesion findings may not represent total disease burden.
In autoimmune disease, uptake is correlated with disease progression, providing a mechanistic assessment of complicated disease etiology. Paired pre/post-imaging suggest that on-treatment activation can be quantified as changes over time.[III]Predictive/association claims. Any references to “prediction,” “correlation,” “association,” “distinguishing responders,” or similar are exploratory and hypothesis-generating unless validated prospectively with predefined thresholds and outcomes.
See figure captions for details on N, criteria, timing, and statistics; methods and limitations are provided below.[VI]Study details govern. See figure captions and cited publications for study design, N, inclusion criteria, comparators, timing, endpoints, statistics, and limitations; those details control in case of discrepancies.

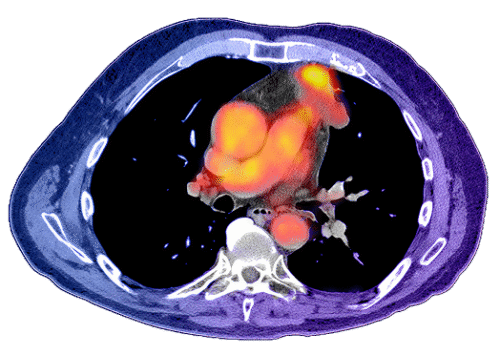
Granzyme B PET Imaging Data in Melanoma
Melanoma (Lesion-level uptake vs. best overall response). Exploratory analysis showing on-treatment CSB-321 signal in lesions that later met response criteria; non-responding lesions tended to remain low.[III]Predictive/association claims. Any references to “prediction,” “correlation,” “association,” “distinguishing responders,” or similar are exploratory and hypothesis-generating unless validated prospectively with predefined thresholds and outcomes.
Granzyme B PET Imaging Data in Non Small Cell Lung Cancer (NSCLC)
Non Small Cell Lung Cancer (NSCLC) (Dynamic analysis of CSB-321). Dynamic analysis of PET tracer uptake reveals stable and differential accumulation between responders and non-responders.[III]Predictive/association claims. Any references to “prediction,” “correlation,” “association,” “distinguishing responders,” or similar are exploratory and hypothesis-generating unless validated prospectively with predefined thresholds and outcomes.
Are you an Academic Investigator?
Granzyme B PET Imaging Data in Colitis
Colitis (Inflammatory bowel disease characterization). CSB-321 highlights immune activation in models of inflammatory bowel disease as well as the ability to accurately reflect disease modulation following therapeutic intervention.[VII]Preclinical limitations. Animal, ex vivo, and other preclinical findings may not predict clinical outcomes in humans; results can differ by species, model, dosing, and study design.
Granzyme B PET Imaging Data in Combination Therapy
Combination Therapy (pre vs. post add-on). Comparisons of standard and novel IO combination approaches in animal models support the ability to distinguish individual response as well as compare therapeutic efficacy across treatment groups.[VII]Preclinical limitations. Animal, ex vivo, and other preclinical findings may not predict clinical outcomes in humans; results can differ by species, model, dosing, and study design.
Granzyme B PET Imaging Data in Inflammatory Lung Disease
Inflammatory Lung Disease (specificity context). Animal models of inflammatory lung disease and subsequent treatment support the ability to differentiate inflammatory from fibrotic idiopathic lung disease.[VII]Preclinical limitations. Animal, ex vivo, and other preclinical findings may not predict clinical outcomes in humans; results can differ by species, model, dosing, and study design. Pre-publication available upon request.
Radiotherapeutic Data
In models, CSB-421 accumulated at granzyme B–rich interfaces and delivered localized radiotherapy, resulting in a 100% complete response compared to controls, with no damage to healthy cells; details of off-target exposure and tolerability are provided in the study.[VII]Preclinical limitations. Animal, ex vivo, and other preclinical findings may not predict clinical outcomes in humans; results can differ by species, model, dosing, and study design.
Exploratory sequencing suggests a potential feed-forward effect: initial dosing may recruit additional effector cells, increasing future docking sites.[I]Forward-looking statements. Statements about potential benefits, future studies, timelines, or development plans are forward-looking and subject to risks and uncertainties; actual results may differ.
Studies are ongoing; no claims of safety or efficacy are made.[II]Early/ongoing human research. Human data referenced are from early, exploratory, or ongoing studies and may be subject to change as additional data become available. See figure captions for N, comparators, timing, endpoints, and statistics.[VI]Study details govern. See figure captions and cited publications for study design, N, inclusion criteria, comparators, timing, endpoints, statistics, and limitations; those details control in case of discrepancies.

CSB-421 Radiotherapeutic Data in Colon Cancer
CSB-421 (preclinical) in colon cancer models: In CT26 and MC38 murine tumors, a granzyme B–targeted theranostic strategy (imaging with ^68Ga-GZP; therapy with ^90Y-GZP) given after immune checkpoint inhibition produced a dose-dependent anti-tumor effect, with the high-dose ^90Y-GZP arm achieving complete tumor response and improved survival versus controls.[VII]Preclinical limitations. Animal, ex vivo, and other preclinical findings may not predict clinical outcomes in humans; results can differ by species, model, dosing, and study design. Body-weight tracking and organ histology showed no systemic toxicity signals in the reported groups.[VIII]Radiation safety/dosimetry. Radiopharmaceuticals involve exposure to ionizing radiation. Dosimetry, off-target exposure, and long-term risks are the subject of ongoing evaluation. Click the chart for dosing, timing, N, endpoints, and Methods; full study details in Pharmaceutics (2022).[VI]Study details govern. See figure captions and cited publications for study design, N, inclusion criteria, comparators, timing, endpoints, statistics, and limitations; those details control in case of discrepancies.

-
I
Forward-looking statements. Statements about potential benefits, future studies, timelines, or development plans are forward-looking and subject to risks and uncertainties; actual results may differ.
-
II
Early/ongoing human research. Human data referenced are from early, exploratory, or ongoing studies and may be subject to change as additional data become available.
-
III
Predictive/association claims. Any references to “prediction,” “correlation,” “association,” “distinguishing responders,” or similar are exploratory and hypothesis-generating unless validated prospectively with predefined thresholds and outcomes.
-
IV
Investigational status. All CytoSite Bio product candidates (including CSB-321 and CSB-421) are investigational; they are not approved for sale. Safety and efficacy have not been established.
-
V
Lesion-level heterogeneity. Lesion-level readouts can vary within the same patient; single-lesion findings may not represent total disease burden.
-
VI
Study details govern. See figure captions and cited publications for study design, N, inclusion criteria, comparators, timing, endpoints, statistics, and limitations; those details control in case of discrepancies.
-
VII
Preclinical limitations. Animal, ex vivo, and other preclinical findings may not predict clinical outcomes in humans; results can differ by species, model, dosing, and study design.
-
VIII
Radiation safety/dosimetry. Radiopharmaceuticals involve exposure to ionizing radiation. Dosimetry, off-target exposure, and long-term risks are the subject of ongoing evaluation.
Need lesion-level clarity on immune engagement?
CSB-321 PET (investigational) visualizes extracellular granzyme B to map within-patient heterogeneity.
